The new GCSE for Science (and Physics) requires pupils to discuss Energy in a new way. I was very dubious about this new speak energy model at first but having introduced it with 7 classes since September I can say I am mildly converted to it. I think it’s a better way describe much of what goes on in the universe than the older model for energy. Like most models we use in Physics to explain physical processes it is not perfect. But you are required to use it. So here goes my attempt at it.
THE MODEL
Firstly, there is no such a thing as energy. I don’t use joules of energy to walk
up the stairs -
The old model for energy contained 9 (or 10) types/forms of energy and that energy can be transferred from any type to any other type depending on the process being described. We gave these types equations which explained how they worked and what we had to do to increase or decrease them.
Behind this old model was the universal principle of conservation of energy which states that energy cannot be created or destroyed and that is only changes from type to type.
The new model for energy is also based on the principle of conservation of energy so that is the same. The major differences come from how we describe different types of energy. We now say that an object can have joules of energy in one of its 8 possible stores and that to transfer these joules of energy from one store to another it has to be done by one of 4 pathways.
Part of my problem with this new model was where sound and light fits into the picture. Light is now a transfer of energy rather than a measure of joules itself. Sound is where I disagree with some textbooks. Sound is listed in the radiation transfer but I think it belongs more in the mechanical transfer as sound is the mechanical movement of particles.
USING THE MODEL
As with the older model you will be required to explain real-
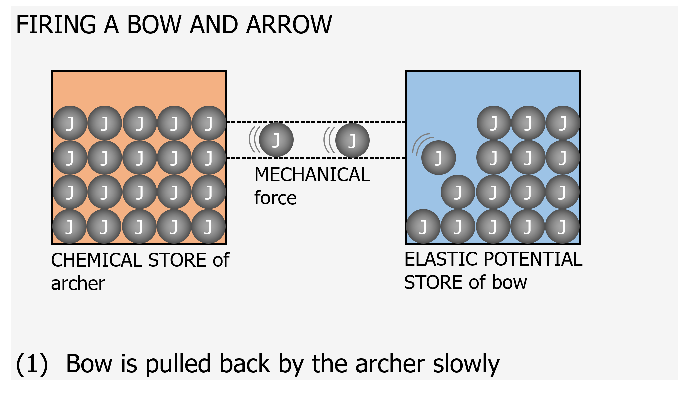
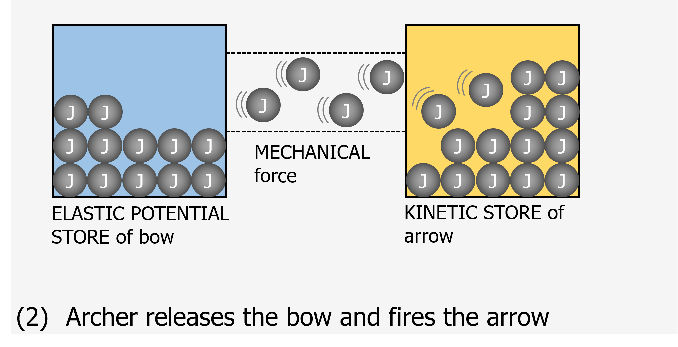
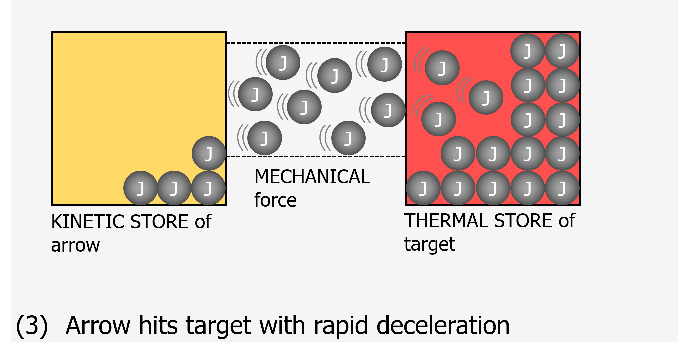
I think of a joule now as being like a marble. This marble can be located in one of the 8 stores at any one time. What we would have traditionally thought of as a transfer of energy should now be visualised as the marble (joule) moving from one container (store) to another one container (store) through a newly created pipe (pathway). This pathway is generated when a force is exerted. The nice thing about this visualisation is that the containers and pipe can be any size or width as it represents real life.
EXAMPLE
A container for the kinetic energy store for a small mass might be small and it fills up with marbles (joules) as it increases speed until it reaches some maximum capacity for kinetic energy. If this object were to suddenly hit a target and stop almost instantaneously then there would be a large transfer of joules very quickly to the thermal stores of the target. The pipe (pathway) for this would be very wide as lots of joules will have been transferred in a short time. In time this target will cool down and transfer joules to the thermal stores of the environment. This pathway may be smaller to represent the slower cooling time for the target than its heating up time. The thermal stores of the environment could be considered as a massive store which doesn’t really get that warm even when lots of joules are transferred to it as these joules will be widely spread out.
VISUALISING STORES AND PATHWAYS